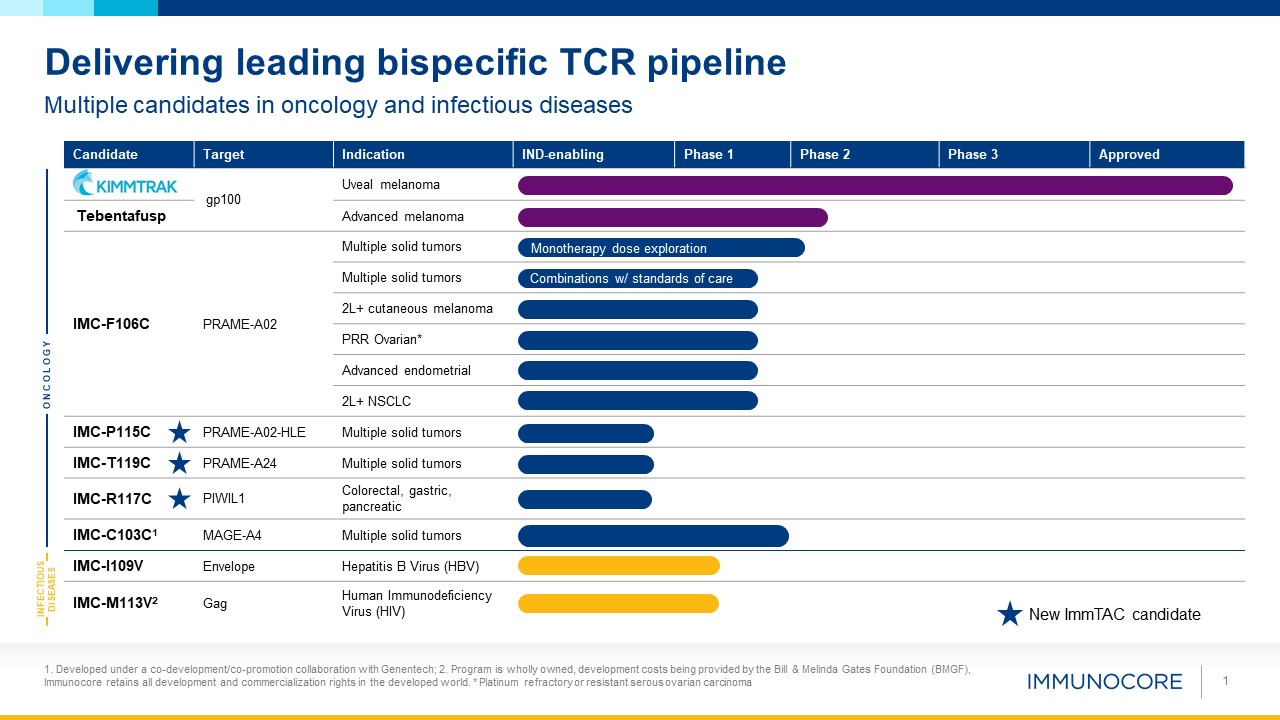
1 Multiple candidates in oncology and infectious diseases Delivering leading bispecific TCR
pipeline Candidate Target Indication IND-enabling Phase 1 Phase 2 Phase 3 Approved gp100 Uveal melanoma Tebentafusp Advanced melanoma IMC-F106C PRAME-A02 Multiple solid tumors Multiple solid tumors 2L+ cutaneous melanoma PRR
Ovarian* Advanced endometrial 2L+ NSCLC IMC-P115C PRAME-A02-HLE Multiple solid tumors IMC-T119C PRAME-A24 Multiple solid tumors IMC-R117C PIWIL1 Colorectal, gastric, pancreatic IMC-C103C1 MAGE-A4 Multiple solid
tumors IMC-I109V Envelope Hepatitis B Virus (HBV) IMC-M113V2 Gag Human Immunodeficiency Virus (HIV) ONCOLOGY INFECTIOUS DISEASES Monotherapy dose exploration Combinations w/ standards of care New ImmTAC candidate 1. Developed under
a co-development/co-promotion collaboration with Genentech; 2. Program is wholly owned, development costs being provided by the Bill & Melinda Gates Foundation (BMGF), Immunocore retains all development and commercialization rights in the
developed world. * Platinum refractory or resistant serous ovarian carcinoma